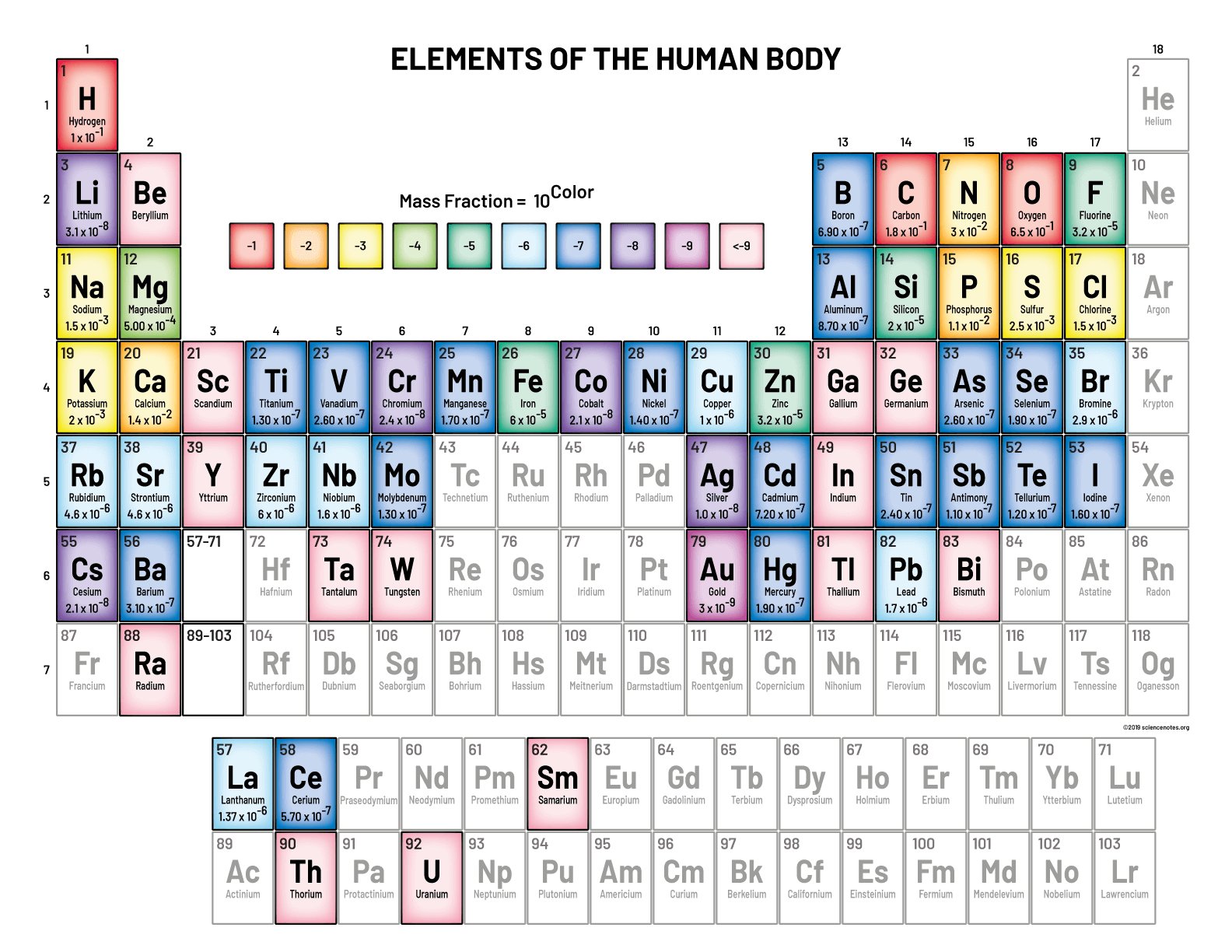
Elements in the human body typically reside in specific locations on the periodic table. Oxygen, the most abundant element in the human body, can be found in group 16. Calcium, crucial for bone health, is located in group 2. Iron, essential for oxygen transport, belongs to group 8. Understanding where do the elements in the human body tend to be located on the table provides insights into our biological composition. Let’s explore this fascinating connection between chemistry and our bodies.
Where Do the Elements in the Human Body Tend to Be Located on the Table
Welcome, young scientists, to an exciting journey into the world of elements and their role in our amazing human bodies! Have you ever wondered where the different elements in your body can be found on the periodic table? Let’s dive in and explore the fascinating world of elements and their locations within our bodies.
The Basic Building Blocks: Carbon, Hydrogen, Oxygen, and Nitrogen
Our bodies are made up of various elements, with four main elements – carbon, hydrogen, oxygen, and nitrogen – making up the majority of our body’s composition. These elements can be found in different groups on the periodic table.
Carbon, known for its versatility, is found in Group 14 of the periodic table. It plays a crucial role in forming the backbone of organic molecules, such as proteins, fats, and carbohydrates, which are essential for our body’s functions.
Hydrogen, the lightest element, resides in Group 1 of the periodic table. It is a key component of water (H2O) and is also present in various organic compounds within our bodies.
Oxygen, a vital element for respiration, is located in Group 16 on the periodic table. It is a part of water and is essential for the process of generating energy in our cells through respiration.
Nitrogen, found in Group 15, is crucial for building proteins and nucleic acids, such as DNA and RNA, which are responsible for carrying genetic information and performing essential functions within our bodies.
Trace Elements: Iron, Calcium, Sodium, and Potassium
In addition to the major elements, our bodies also require trace elements in smaller amounts to function properly. Let’s explore where some of these essential trace elements can be found on the periodic table.
Iron (Fe)
Iron, located in Group 8 of the periodic table, is a crucial element for carrying oxygen in our blood and supporting various metabolic processes in the body. It is a key component of hemoglobin, the protein responsible for transporting oxygen from the lungs to the body’s tissues.
Calcium (Ca)
Calcium can be found in Group 2 on the periodic table. It plays a vital role in building and maintaining strong bones and teeth, as well as supporting muscle function, nerve signaling, and blood clotting within our bodies.
Sodium (Na) and Potassium (K)
Sodium and potassium, both located in Group 1 of the periodic table, are essential for maintaining proper fluid balance, nerve function, and muscle contractions in our bodies. While sodium is often associated with table salt, potassium is abundant in fruits and vegetables.
Transition Metals: Zinc, Copper, and Magnesium
Transition metals are a group of elements that have unique properties and are essential for various biological processes in our bodies. Let’s explore where some of these important transition metals can be found on the periodic table.
Zinc (Zn)
Zinc, located in Group 12, is a vital element for immune function, wound healing, and DNA synthesis within our bodies. It is also involved in taste perception and hormone regulation.
Copper (Cu)
Copper, found in Group 11, is important for the formation of red blood cells, energy production, and maintaining healthy connective tissues within our bodies. It also acts as an antioxidant, protecting our cells from damage.
Magnesium (Mg)
Magnesium can be found in Group 2 of the periodic table. This essential mineral is involved in over 300 biochemical reactions in the body, including muscle and nerve function, energy production, and bone health.
Rare Earth Elements: Cerium, Lanthanum, and Gadolinium
While rare earth elements are not commonly discussed in the context of human biology, they are still present in trace amounts in our bodies and play specific roles. Let’s take a quick look at where some of these rare earth elements can be located on the periodic table.
Cerium (Ce)
Cerium, a rare earth element found in the lanthanide series, has applications in catalytic converters and glass production. While it is not considered essential for human health, cerium can still be found in trace amounts in the body.
Lanthanum (La) and Gadolinium (Gd)
Lanthanum and gadolinium, also part of the lanthanide series, have industrial applications and are used in various technologies. While they are not known to have specific biological functions in the human body, they may still be present in trace amounts due to environmental exposure.
Elementary scientists, we’ve journeyed through the periodic table and explored where the elements in the human body tend to be located. From the basic building blocks of carbon, hydrogen, oxygen, and nitrogen to the essential trace elements and transition metals that support our body’s functions, each element plays a unique role in maintaining our health and well-being.
Remember, young scientists, our bodies are a complex and wondrous combination of elements that work together harmoniously to keep us healthy and strong. Keep exploring the world of science and stay curious about the fascinating elements that make up our incredible human bodies!
Stay curious, stay excited, and keep exploring the amazing world of science!
What's in Your Body 60 Elements! #HumanBody #ElementsInTheBody #BodyChemistry #PeriodicTable #Scien
Frequently Asked Questions
Which elements in the human body are typically found in Group 1 of the periodic table?
Elements in Group 1 of the periodic table, such as sodium and potassium, are commonly found in the human body. Sodium plays a crucial role in maintaining fluid balance, while potassium is essential for nerve function and muscle contractions.
What are some elements from Group 7 that are essential for human health?
Elements in Group 7, like chlorine and iodine, are vital for human health. Chlorine is important for digestive processes, and iodine is crucial for thyroid hormone production, which regulates metabolism.
How do elements from Group 14 contribute to the structure of the human body?
Elements in Group 14, such as carbon and silicon, play key roles in the structure of the human body. Carbon is the building block of organic molecules like proteins and DNA, while silicon contributes to the strength of bones and connective tissues.
Final Thoughts
In conclusion, the elements in the human body tend to be located on the periodic table according to their properties and functions. For instance, essential elements like oxygen and carbon, crucial for life, are found in the top left corner. Minerals like calcium and phosphorus, vital for bone health, are located towards the bottom. Understanding where do the elements in the human body tend to be located on the table helps us appreciate the significance of each element in maintaining health and balance within the body.